Research News
Bringing the Hospital Home
UHCOP Professor Developing Novel Model of Skin Necrosis for Potential New At-home Subcutaneous Antibiotic Delivery System
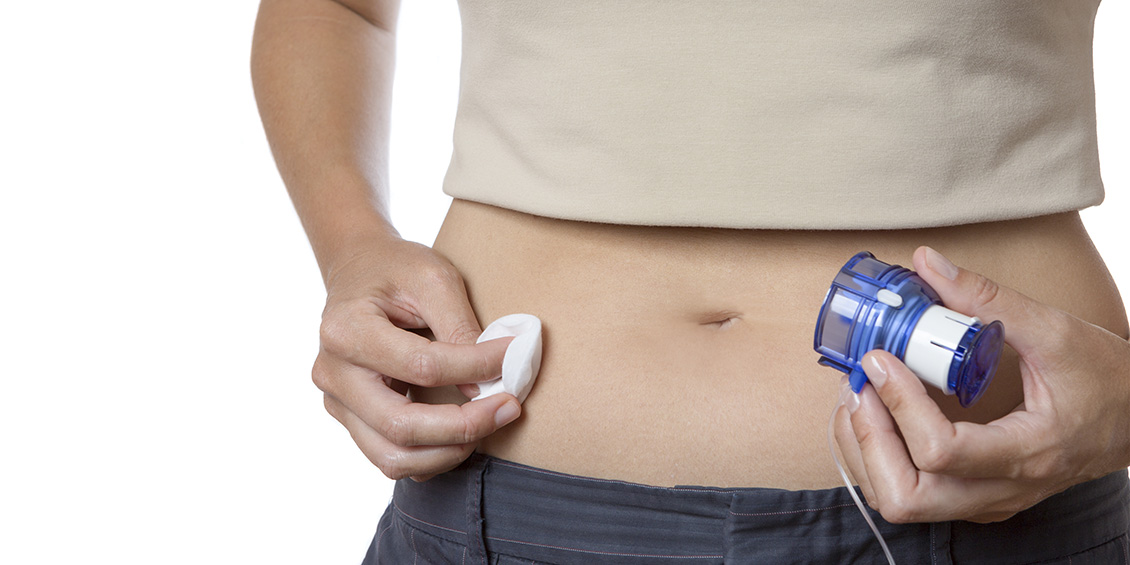
As the health care industry continues to seek alternative treatment models to simultaneously reduce costs and inconvenience to patients, UH College of Pharmacy Professor and infectious diseases researcher Vincent H. Tam, Pharm.D., BCPS (AQ-ID), is developing a pre-clinical model of skin necrosis in preparation for clinical trials on a new in-home infusion platform for antibiotic therapy.
Supported by a $546,700 research grant from Massachusetts-based scPharmaceuticals Inc., the project aims to establish key parameters – including concentration, volume and duration – for a new home-based subcutaneous administration of an antibiotic therapy instead of the intravenous (IV) administration in a hospital setting currently available and approved by the Food & Drug Administration.
"When you have patients undergoing therapy with drugs that have low oral absorption, typically these patients have to be managed in the hospital," Tam said. "The concept of that kind of care is changing to the point where the patient doesn’t have to be institutionalized for a long period of time just because you need an IV antibiotic. Generally, people don’t like to deal with an IV if they have a choice. You’re much more likely to get sepsis from an infection from an IV, and you have to have surgery to place those accesses – so you may be trading one problem for another."
Services as chemotherapy and even dialysis treatments were once only available on-site at a hospital, but those services are now routinely available in outpatient centers. Tam said some home-based infusion therapies already are available today, but with the development of better, safer technologies and drug formulations, the market for such services is poised for significant expansion.
"(Subcutaneous infusion therapy) is not necessarily risk-free, but there is precedent: People have been doing self-injections of insulin on a daily basis for the rest of their lives and are fine," Tam said. "But, a common problem of giving drugs under the skin in general is local inflammation, especially if it’s puncturing the skin daily. You can have local irritation and necrosis, so obviously this is a concern that manufacturers, patients and caregivers have – and we need to have an idea of how to address this problem."
Skin – the largest organ in the human body – presents unique challenges for pharmaceutical companies and device manufacturers developing new products, particularly in the pre-clinical phase that can make or break a product’s progression and the associated costs of moving through the FDA review/approval process.
"There’s no widely established way of dealing with this problem in a systematic, scientific fashion," Tam said. "In a preclinical model, you can’t really use cell lines because the skin is a heterogeneous organ; there is no one definitive cell line that can represent the skin structure, so we are somewhat obligated to use animal models to help us.
"The whole point is that you don’t want to try this out on humans, without knowing or having high degree of confidence of the likely outcomes. We’re aiming for maximum patient convenience with the minimum local intolerance; my hypothesis is that there is a sweet spot somewhere in the middle."
Tam’s project collaborators are fellow UHCOP Professor and infectious diseases researcher Kevin W. Garey, Pharm.D., M.S., FASHP, and Baylor College of Medicine Assistant Professor and dermatopathologist Daniel N. Cohen, M.D, Ph.D., F.C.A.P.