Faculty Profile
 Stuart E. Dryer
Stuart E. Dryer
Moores Professor
Department of Biology and Biochemistry
Research Division: Cell and Molecular Biology (Primary)
Office: Science & Research 2, 242E
Contact: sdryer@uh.edu - 713-743-2697
Education: Ph.D., Saint Louis University
Dr. Stuart Dryer’s laboratory studies physiological and pathophysiological processes that occur in the kidney. The current research focuses on the role of ion channels and receptors in the regulation of cells that form the glomerular filtration barrier, and the proximal tubule. The lab has recently focused on a Ca2+-permeable cation channel known as TRPC6. Mutations that cause gain of function in these channels lead to devastating genetic forms of kidney disease. Dryer’s lab is interested in understanding the normal function and regulation of TRPC6 channels, including the factors that activate their gating and abundance at the cell surface. They are also interested in the role of TRPC6 in driving non-genetic forms of glomerular disease.
In a second research program, he has been studying renal NMDA receptors and their role in normal and abnormal physiology. NMDA receptors are best known for their role in synaptic transmission. However, they are also present in many peripheral tissues including the kidney, where their function remains a mystery. The Dryer Lab has recently shown that sustained activation of NMDA receptors in podocytes induces oxidative stress, changes in expression of regulatory proteins, and apoptotic cell death. They have also observed marked up-regulation of NMDA receptors throughout the kidney in mouse models of type 1 diabetes, and have shown that sustained treatment of NMDA antagonists reduces nephropathy in mouse models of type 1 diabetes. They are currently working to understand how NMDA receptors are regulated in normal and diseased kidneys, including understanding the molecules that normally cause them to become active.
- Kim, E. Y. and Dryer, S.E. (2021). RAGE and αVβ3-integrin are essential for suPAR signaling in podocytes. Biochim Biophys Acta Mol Basis Dis. 1867:166186.
- Kim, E. Y. and Dryer, S.E. (2021). Effects of TRPC6 inactivation on glomerulosclerosis and renal fibrosis in aging rats. Cells 10: 856
- Dryer, S. E., Roshanravan, H., and Kim, E. Y. (2019). TRPC channels: Regulation, dysregulation and contributions to chronic kidney disease. Biochim Biophys Acta Mol Basis Dis. 1865:1041-1066.
- Kim, E. Y., Yazdizadeh Shotorbani, P., & Dryer, S. E. (2019). Trpc6 inactivation does not affect loss of renal function in nephrotoxic serum glomerulpnephritis in rats, but reduces severity of glomerular lesions. Biochemistry and Biophysics Reports. 17:139-150.
- Kim E, Y., Hassanzadeh Khayyat, N., and Dryer, S. E. (2018). Mechanisms underlying modulation of podocyte TRPC6 channels by suPAR: Role of NADPH oxidases and Src family tyrosine kinases. Biochimica Biophysica Acta-Molecular Basis of Disease 1864: 3527-3536.
- Dryer, S. E. and Kim, S. E. (2018). Permeation and rectification in canonical transient receptor potential-6 (TRPC6) channels. Frontiers in Physiology 9:1055.
- Kim, E. Y., Yazdizadeh Shotorbani, P., & Dryer, S. E. (2018). Trpc6 inactivation confers protection in a model of severe nephrosis in rats. Journal of Molecular Medicine (Berlin). 96: 631-644.
- Kim, E. Y., Roshanravan, H., & Dryer, S. E. (2017). Changes in podocyte TRPC channels evoked by plasma and sera from patients with recurrent FSGS and by putative glomerular permeability factors. Biochimica Biophysica Acta-Molecular Basis of Disease. 1863: 2342-2354.
- Roshanravan, H., Kim, E.Y., & Dryer, S. E. (2016). NMDA receptors as potential therapeutic targets in diabetic nephropathy: Increased renal NMDA receptor subunit expression in Akita mice and reduced nephropathy following sustained treatment with memantine or MK-801. Diabetes 65: 3139-50.
- Kim, E. Y., Roshanravan, H., and Dryer, S. E.* (2015) Syndecan-4 ectodomain evokes mobilization of podocyte TRPC6 channels and their associated pathways: An essential role for integrin signaling. Biochimica Biophysica Acta – Molecular Cell Research. 1853: 2610-20
- Dryer, S. E. (2015). Glutamate receptors in the kidney. Nephrology Dialysis Transplantation 30: 1630-8.
- Roshanravan, H. and Dryer, S.E. (2014). ATP acting through P2Y receptors causes activation of podocyte TRPC6 channels: Role of podocin and reactive oxygen species. American Journal of Physiology-Renal Physiology 306: F1088-97.
- Wilson, C. and Dryer, S. E. (2014). A mutation in TRPC6 channels abolishes their activation by hypoosmotic stretch but does not affect activation by diacylglycerol or G protein signaling cascades. American Journal of Physiology-Renal Physiology 306: F1018-25
- Hagmann, H., Kuczkowski, A., Ruehl, M., Lamkemeyer, T., Brodesser, S., Horke, S., Dryer, S. E., Schermer, B., Benzing, T., Brinkkoetter, P. T. (2014). Breaking the chain at the membrane: paraoxonase 2 counteracts lipid peroxidation at the plasma membrane. FASEB Journal 28: 1769-79
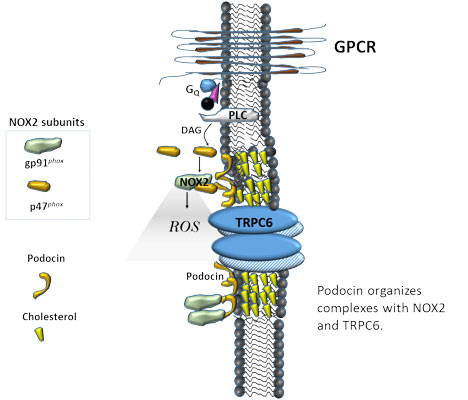
- Kim E. Y., Anderson, M., Wilson, C., Hagmann, H., Benzing, T. & Dryer, S. E. (2013). NOX2 interacts with podocyte TRPC6 channels and contributes to their activation by diacylglycerol: Essential role of podocin in formation of this complex. American Journal of Physiology-Cell Physiology 305: C960-71.
- Anderson, M., Kim E. Y., Hagmann, H., Benzing, T. & Dryer, S. E. (2013). Opposing effects of podocin on the gating of podocyte TRPC6 channels evoked by membrane stretch or diacylglycerol. American Journal of Physiology-Cell Physiology 305: C276-89.
- Kim, E. Y., Anderson. M. & Dryer, S. E. (2012). Insulin increases surface expression of TRPC6 channels in podocytes: An essential role for NADPH oxidases and reactive oxygen species.
National Honors and Awards
Fellow, American Association for the Advancement of Science
Fellow, American Society of Nephrology
Current and Past Grant Review Panel Memberships
National Institutes of Health
American Diabetes Association
Editorial Boards
Biochmica Biophysica Acta- Molecular Basis of Disease
PLoS ONE
Frontiers in Physiology
Frontiers in Molecular Neuroscience
Bioelectricity